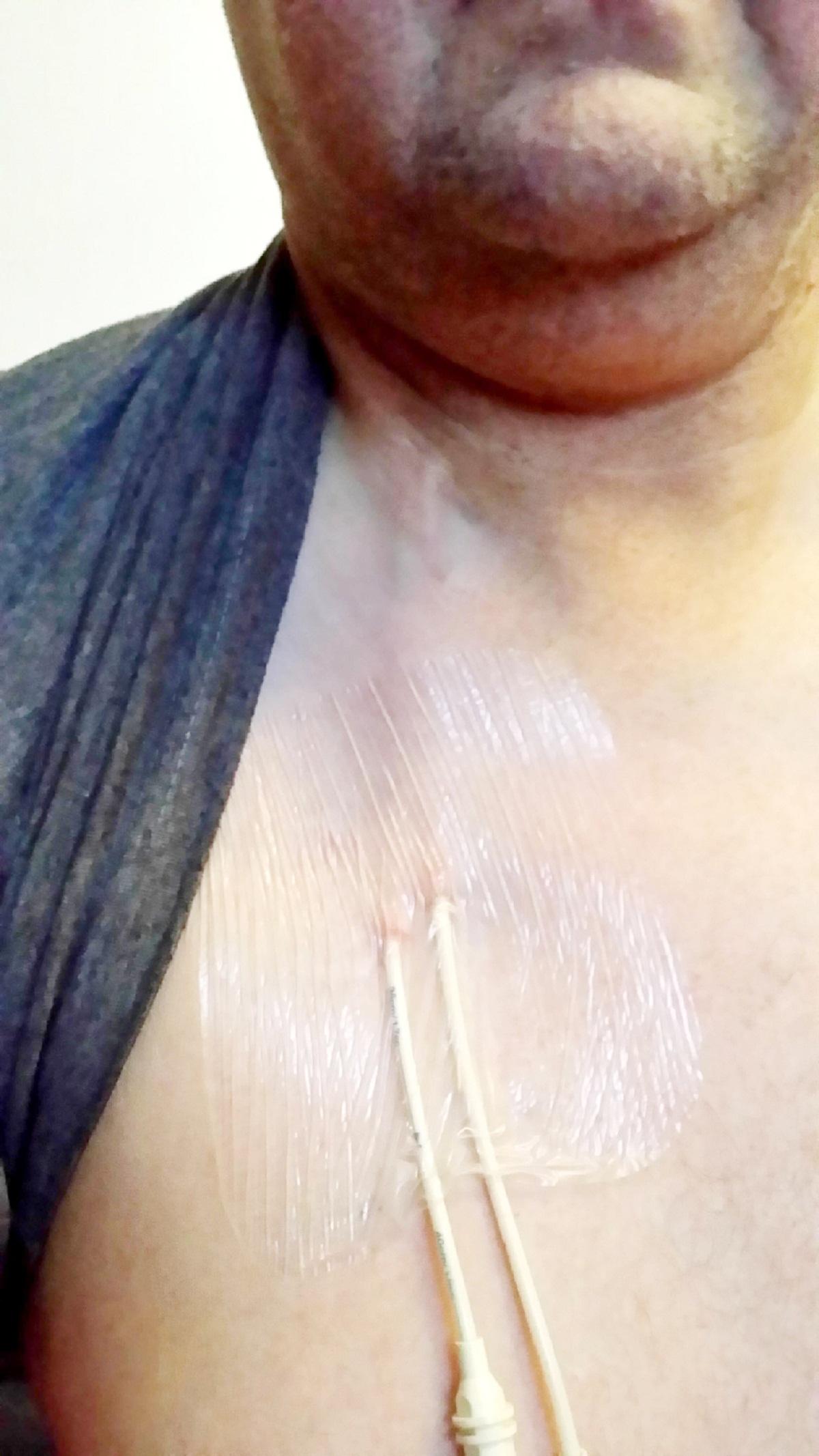
A CANCER patient who was weeks away from death has made medical history after becoming the first person in Europe to trial a groundbreaking drug.
Doctors thought Ian Brooks, aged 47, only had weeks to live after exhausting nearly all treatment options for an aggressive form of non-hodgkin’s lymphoma, which had left his body full of tumours.
When Mr Brooks was asked if he wanted to trial the new drug — Brentuximab Vedotin — he volunteered immediately in bid cure his cancer and “help other people”.
But doctors at the Christie never expected the treatment to have such rapid and “remarkable” results in tackling the tumours.
Mr Brooks, of Sharnford Close, The Haulgh, is now in remission and the drug has been made available for the NHS patients with the same rare condition.
He said: “When you’re told you have cancer, you imagine you have one or two tumours, but it turned I had them all over my body.
“When they asked me about the trial, I just thought I had nothing to lose at the stage. To be truthful, I am not a selfish person.
"I thought if it went well, it could boost my chance of survival and if it didn’t and I died, they could use me as a medical study.
“Whatever happened, taking part would at least benefit other people in the future.
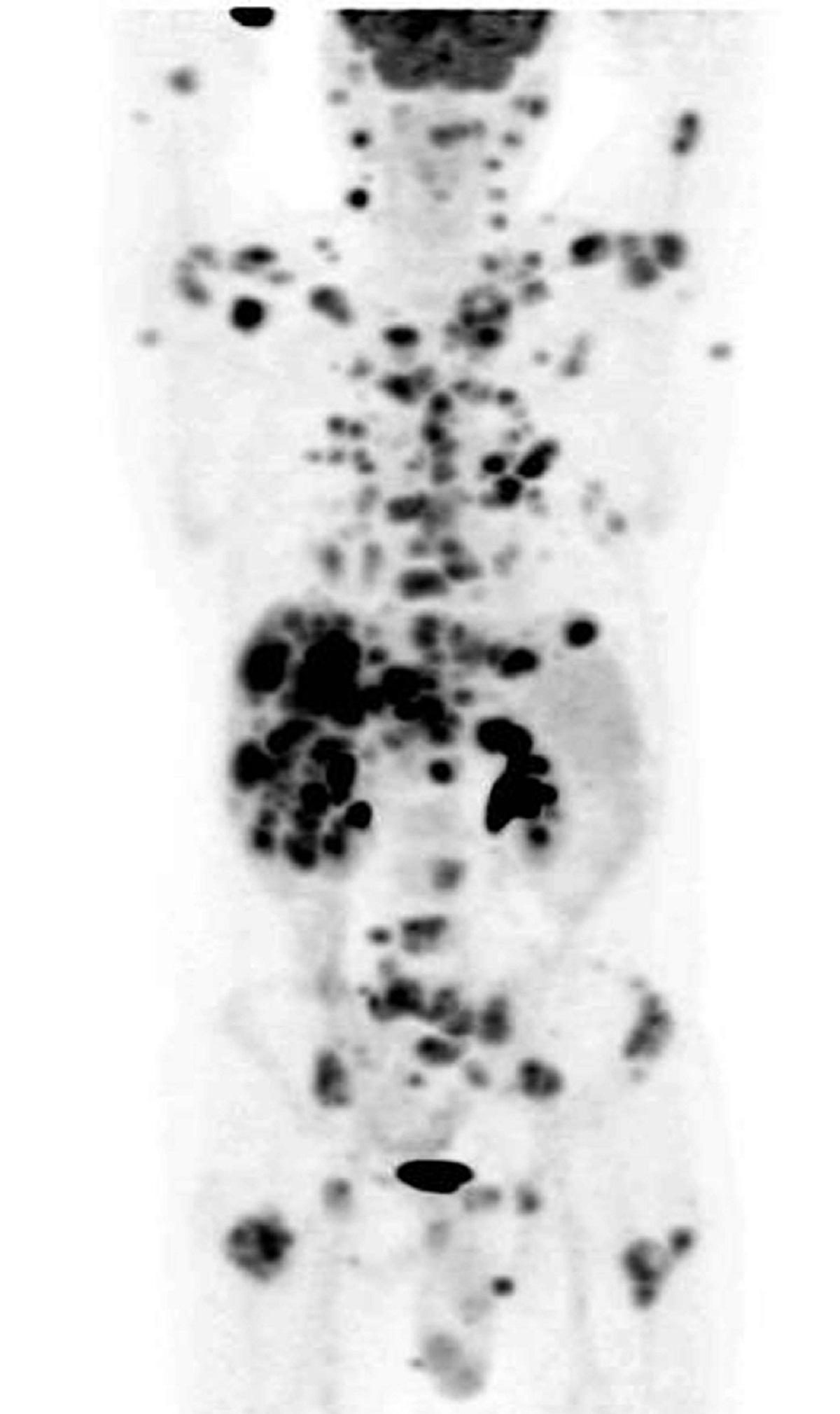
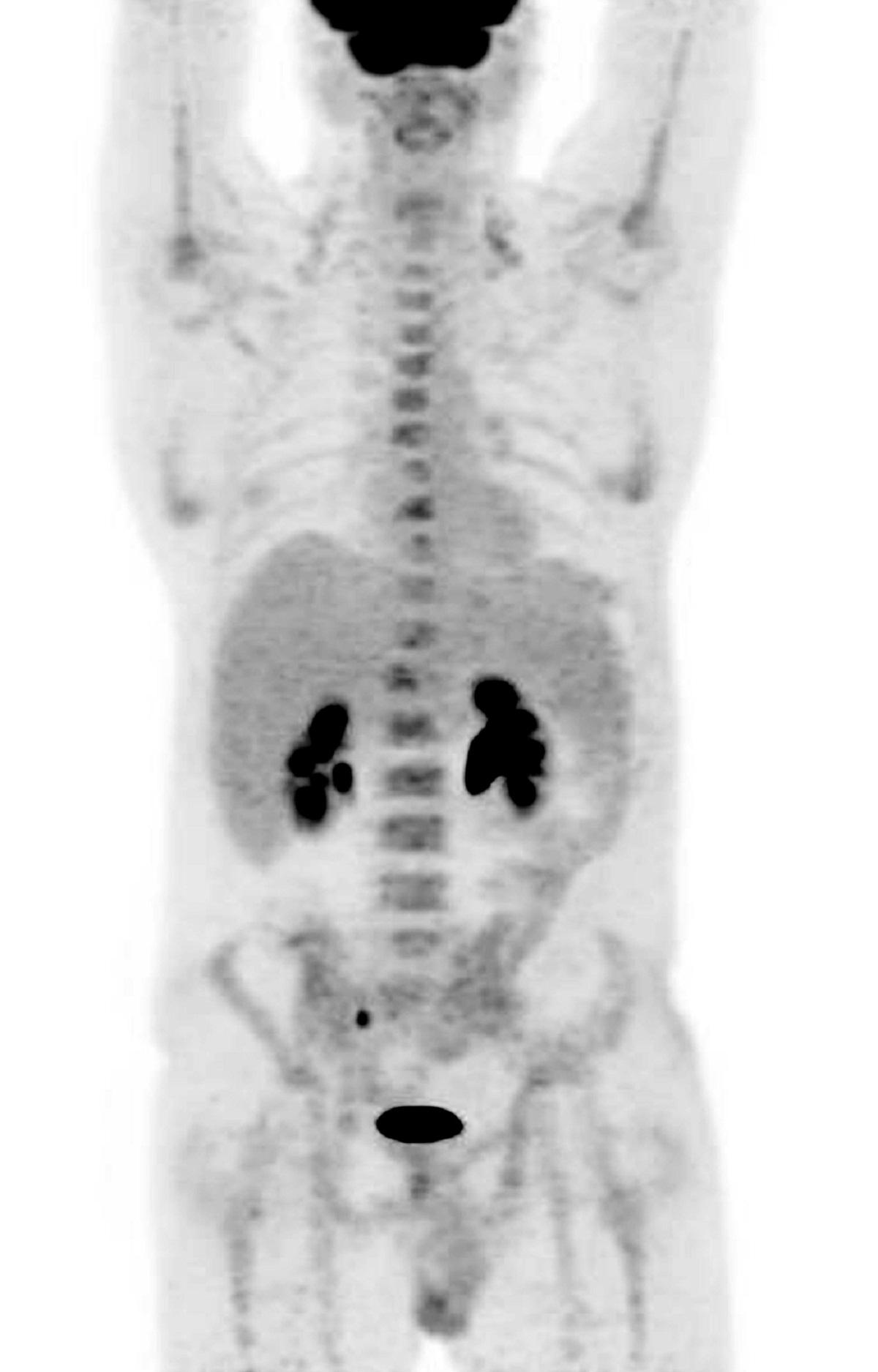
“When I’d had the treatment and they’d showed me the scans, I couldn’t believe the difference. I could see that the tumours had gone.”
Mr Brooks was first diagnosed with Anaplastic Large Cell Lymphoma in 2001, aged 31, and responded well to treatment.
But in 2008 the cancer returned and he underwent a stem cell transplant.
Yet when Mr Brooks found a lump in his groin a year later, doctors confirmed the cancer had returned.
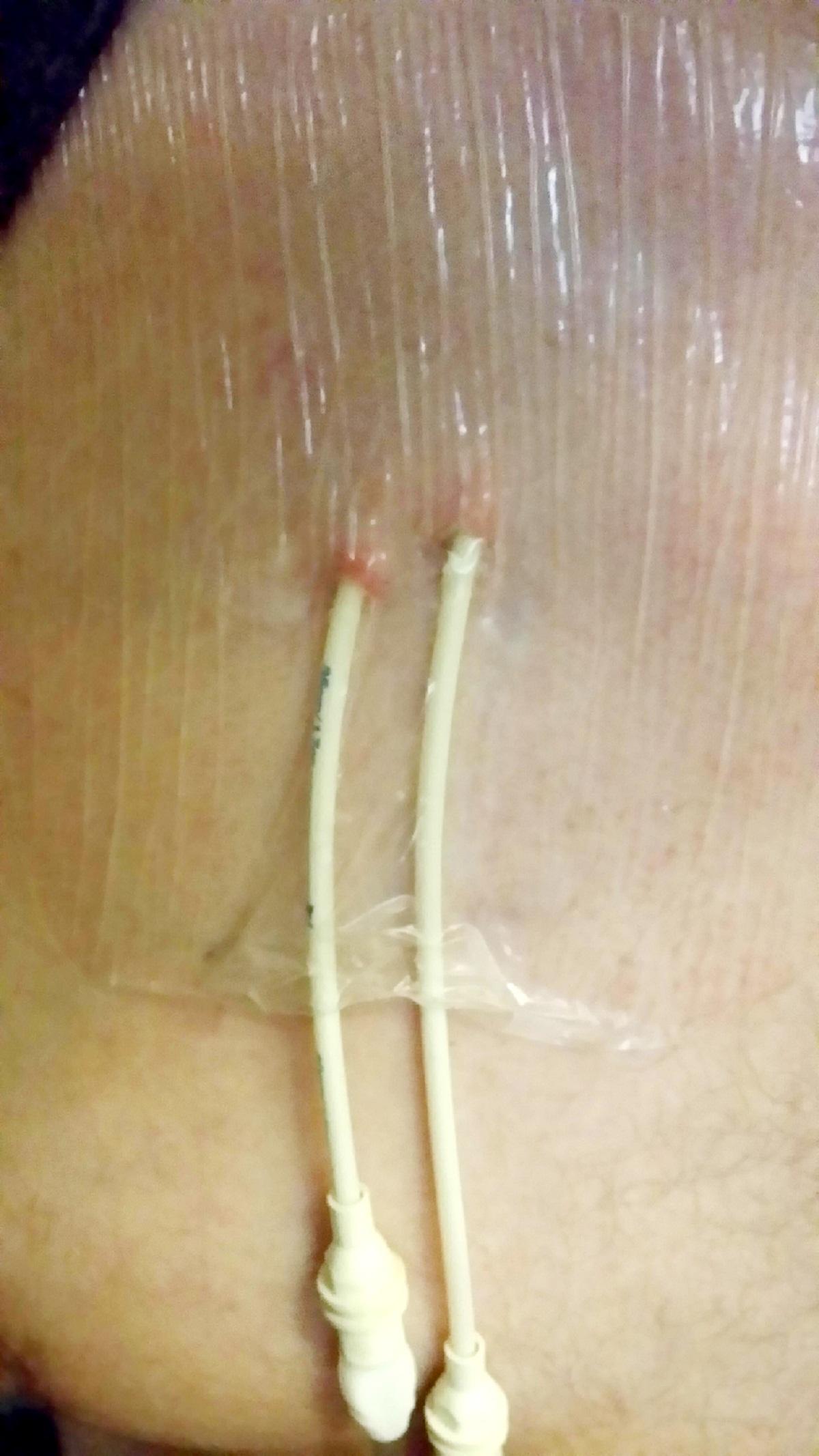
When he volunteered to trial the drug — known at the time simply as SGN-35 — he was put on a 12 week course of treatment.
The drug is described as a “targeted treatment” that locks only to the lymphoma cells and delivers a toxin or poison directly — unlike chemotherapy, which tackles all cells in the body.
Dr Adam Gibb, clinical research fellow in lymphoma at the Christie, said: “Ian had exhausted all other areas of treatment and we really thought he only had a few weeks left to live.
“We knew this was an exciting drug but we never expected to see such a dramatic improvement in his condition. The results after 12 weeks were remarkable.
“This is a great example of why taking part in a clinical trial can be so positive for medicine.”





Comments: Our rules
We want our comments to be a lively and valuable part of our community - a place where readers can debate and engage with the most important local issues. The ability to comment on our stories is a privilege, not a right, however, and that privilege may be withdrawn if it is abused or misused.
Please report any comments that break our rules.
Read the rules hereLast Updated:
Report this comment Cancel